Spinal Muscular Atrophy Market is expected to reach US$ 20.06 Billion by 2033 | DataM Intelligence
The Spinal Muscular Atrophy Market is expected to reach at a CAGR of 16.5% during the forecast period 2025-2033.
The Spinal Muscular Atrophy Market is witnessing rapid growth driven by gene therapy advancements, early diagnosis adoption, and increasing investment in rare disease treatment innovation.”
AUSTIN, TX, UNITED STATES, February 27, 2026 /EINPresswire.com/ -- Market Overview:— DataM Intelligence
The Global Spinal Muscular Atrophy Market has witnessed remarkable transformation over the past decade, driven by advancements in genetic therapies, early disease diagnosis, and increased awareness of rare neuromuscular disorders. Spinal muscular atrophy is a rare genetic disease characterized by progressive muscle weakness caused by the degeneration of motor neurons. The growing adoption of innovative disease-modifying therapies, particularly gene replacement and RNA-targeted treatments, has significantly reshaped treatment outcomes and market dynamics. Pharmaceutical innovation, coupled with supportive regulatory frameworks and orphan drug incentives, continues to accelerate market expansion globally.
To Download Sample Report Here: https://www.datamintelligence.com/download-sample/spinal-muscular-atrophy-market
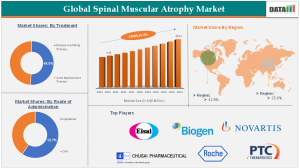
According to DataM Intelligence, The Global Spinal Muscular Atrophy Market was valued at approximately USD 4.40 billion in 2024 and is projected to reach nearly USD 20.06 billion by 2033, growing at a robust CAGR of around 16.5% during the forecast period. Increasing newborn screening programs, improved access to advanced biologics, and rising investments in rare disease research are key growth drivers behind market development. The gene therapy segment currently dominates the market due to its long-term therapeutic benefits and one-time treatment approach. Geographically, North America leads the global SMA market owing to strong healthcare infrastructure, early regulatory approvals, high treatment adoption rates, and significant reimbursement support.
The expansion of precision medicine and personalized healthcare approaches is further strengthening market growth worldwide. Increasing collaborations between biotechnology firms and healthcare institutions are accelerating drug commercialization and improving patient accessibility across emerging economies.
Key Highlights from the Report:
Gene therapy innovations are revolutionizing long-term treatment outcomes in spinal muscular atrophy management.
Rising newborn genetic screening programs are enabling earlier diagnosis and intervention.
Increasing orphan drug approvals are strengthening investment in rare disease therapeutics.
Hospitals remain the leading end-user segment due to specialized treatment requirements.
North America dominates the market due to advanced reimbursement frameworks and clinical research presence.
Expanding clinical pipelines are expected to introduce next-generation SMA therapies during the forecast period.
Market Segmentation:
The Spinal Muscular Atrophy Market is broadly segmented based on treatment type, disease type, route of administration, and end-user. Treatment-based segmentation includes gene therapy, antisense oligonucleotide therapy, and small molecule therapy. Among these, gene therapy has emerged as the fastest-growing segment due to its ability to address the underlying genetic defect responsible for SMA. One-time gene replacement therapies significantly reduce disease progression, improving survival rates and motor function outcomes among pediatric patients.
Antisense oligonucleotide therapies continue to maintain a substantial market share as they help increase survival motor neuron (SMN) protein production. Meanwhile, oral small-molecule therapies are gaining popularity because of improved convenience, patient compliance, and long-term treatment feasibility.
Based on disease type, the market covers Type I, Type II, Type III, and Type IV SMA, with Type I accounting for the largest revenue share due to higher diagnosis rates during infancy and urgent treatment requirements. Early therapeutic intervention remains critical for improving patient prognosis, which supports demand growth within this segment.
From an end-user perspective, hospitals and specialty neurology centers dominate the market as SMA treatments require advanced monitoring, genetic testing, and multidisciplinary clinical expertise. Research institutes and specialty clinics also contribute significantly through ongoing clinical trials and therapy development programs.
Speak to Our Analyst and Get Customization in the report as per your requirements: https://www.datamintelligence.com/customize/spinal-muscular-atrophy-market
Regional Insights:
North America holds the largest share of the spinal muscular atrophy market, supported by favorable healthcare reimbursement policies, strong biotechnology ecosystems, and early adoption of innovative gene therapies. The presence of leading pharmaceutical companies and continuous FDA approvals has accelerated treatment accessibility across the United States and Canada. Additionally, widespread newborn screening initiatives have improved early detection rates, further strengthening regional dominance.
Europe represents the second-largest market, driven by government-funded healthcare systems and growing awareness of rare genetic disorders. Countries such as Germany, France, and the United Kingdom are actively investing in advanced neuromuscular disease treatment programs. Regulatory support for orphan drugs and collaborative research initiatives are enhancing therapeutic availability across the region.
The Asia-Pacific region is expected to witness the fastest growth during the forecast period. Increasing healthcare expenditure, improving genetic diagnostic capabilities, and rising awareness of rare diseases are fueling market expansion in countries such as China, Japan, and India. Government initiatives aimed at improving access to advanced therapies are expected to unlock significant future opportunities.
Latin America and the Middle East & Africa markets are gradually expanding due to improving healthcare infrastructure and increasing partnerships with global pharmaceutical companies. However, limited treatment affordability remains a challenge in these regions.
Market Dynamics:
Market Drivers
One of the primary drivers of the spinal muscular atrophy market is the rapid advancement of gene therapy technologies capable of addressing the root genetic cause of the disease. Increased investment in rare disease drug development, combined with favorable regulatory incentives such as orphan drug designation, has encouraged pharmaceutical companies to accelerate innovation. Growing awareness among healthcare professionals and caregivers regarding early diagnosis further supports treatment adoption. Additionally, expanding newborn screening programs across developed economies significantly enhance early intervention outcomes, boosting overall market demand.
Market Restraints
Despite promising therapeutic advancements, the high cost associated with SMA treatments remains a major restraint for market growth. Gene therapies often involve substantial upfront expenses, creating affordability challenges, particularly in low- and middle-income countries. Limited healthcare reimbursement coverage in developing regions and complex regulatory approval processes also restrict market penetration. Furthermore, logistical challenges related to therapy administration and long-term monitoring add to treatment complexity.
Market Opportunities
The market presents strong opportunities through ongoing research into next-generation therapies and combination treatment approaches. Advances in genetic editing technologies and RNA-based therapeutics are expected to deliver improved efficacy and safety profiles. Expansion into emerging economies through strategic partnerships, patient assistance programs, and government-backed rare disease policies offers untapped growth potential. Digital health integration and remote patient monitoring solutions may also enhance long-term disease management and treatment adherence.
Looking For Full Report? Get it Here: https://www.datamintelligence.com/buy-now-page?report=spinal-muscular-atrophy-market
Frequently Asked Questions (FAQs):
How big is the Global Spinal Muscular Atrophy Market currently?
What is the projected growth rate of the Spinal Muscular Atrophy Market through 2033?
Who are the key players operating in the Global Spinal Muscular Atrophy Market?
What is the market forecast for SMA therapies by the end of the forecast period?
Which region is expected to dominate the spinal muscular atrophy market during the forecast timeline?
Company Insights:
Key companies operating in the Spinal Muscular Atrophy Market include:
F. Hoffmann-La Roche Ltd
Novartis AG
Biogen
Chugai Pharmaceutical Co., Ltd.
PTC Therapeutics, Inc.
Eisai Co., Ltd.
NMD Pharma A/S
Recent Developments:
United States:
January 2026: Newborn screening programs reached 95% coverage in key states, leading to earlier interventions and reduced disease severity.
December 2025: Major reimbursement update by CMS covered combination therapies, boosting access for adult SMA patients nationwide.
November 2025: FDA expanded approval for next-generation gene therapy targeting SMA Type 2 patients, improving motor function outcomes in clinical trials.
Japan:
January 2026: Gene therapy reimbursement fully implemented, with per-capita spend leading Asia-Pacific amid newborn screening mandates.
December 2025: National rare disease initiative funded AI-driven diagnostics, increasing diagnosis rates by 20% in urban centers.
November 2025: MHLW approved antisense oligonucleotide expansion for SMA Type 3, enhancing long-term mobility in prefecture-wide pilots.
Unlock 360° Market Intelligence with DataM Subscription Services: https://www.datamintelligence.com/reports-subscription
Conclusion:
The Spinal Muscular Atrophy Market is undergoing a significant evolution driven by breakthroughs in genetic medicine and precision therapeutics. The shift from symptomatic management toward disease-modifying and potentially curative therapies marks a major milestone in rare disease treatment. Rising awareness, improved diagnostic capabilities, and expanding healthcare investments are expected to sustain strong market growth in the coming years. While treatment affordability and accessibility challenges persist, continuous innovation and supportive healthcare policies are likely to unlock substantial opportunities worldwide. As research advances and patient access improves, the spinal muscular atrophy market is poised to play a transformative role in the future of neuromuscular disease management.
Related Reports:
Neuromuscular Disease Therapeutics Market
Huntington's Disease Market
Sai Kiran
DataM Intelligence 4Market Research
+1 877-441-4866
Sai.k@datamintelligence.com
Visit us on social media:
LinkedIn
X
Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.